SynDaver
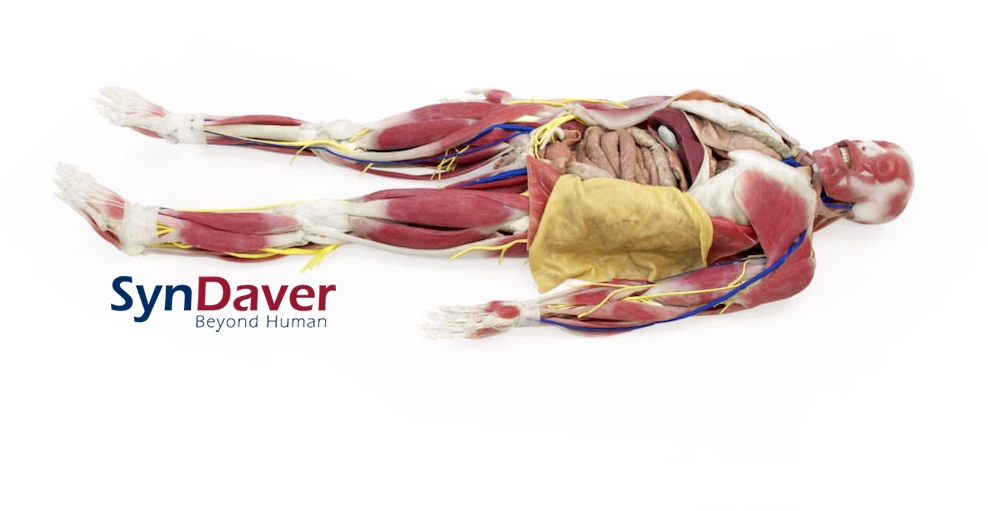
SynDaver Labs is a medical simulation manufacturing company that designs and builds experimentally-validated synthetic human tissues, body parts and human bodies for the purposes of medical education. In providing these products to healthcare simulation programs, the company has developed an extensive catalog of synthetic tissues based on a database of the living tissue properties in humans and animals. This database has been curated over a span of time and is extremely detailed.
Now referred to more simply as SynDaver, the Tampa Florida based company was founded in 2004 to commercialize a novel system of synthetic human body parts for the medical device industry, as well as for academic purposes. SynDaver’s products have served as a means of replicating the human anatomy in great detail, and therefore have provided learners across the globe with an alternative means of clinical experience.
As with many clinical simulation products, this gives learners the ability to practice without the possibility of harming a real life patient. As these patient simulators are made from materials that precisely mimic living tissue, they have been used across a wide range of industries. Industry uses have involved medical device testing and surgical training in high fidelity simulation environments, in addition to consumer product development and crash testing.
Sponsored Content:
For approximately 14 years of business operations, and especially over the most recent four year period, SynDaver Labs has demonstrated strong growth trends. On average, the company has grown by more than 60 percent annually. With this outlook, SynDaver Labs is on track to more than double in size in the years to come.
HealthySimulation.com is dedicated to providing the latest SynDaver news and resources from around the world. To follow along, sign up for our free medical simulation email newsletter, follow @HealthySim on Twitter and @HealthySim on Facebook, or join our HealthySim LinkedIn Group!
SnyDaver strongly credits an executive team for paving the way for this growth in product development and consumer demand. Today, Dr. Christopher Sakezles remains the Founder, CEO, Chief Technology Officer and Chairman of the Board of Directors for SynDaver. The company is also advised by a selected team of experts drawn from the medical device industry, as well as thought leaders in surgical simulation and medical education.
The customer base is broad, diverse, and global. Typical consumers include medical device manufacturers, hospitals, medical schools, and educational institutions (with EMT, nursing, anatomy, veterinary medicine, dentistry, engineering, mortuary science, surgical technician, physical therapy and sports science programs). The company even sells to middle and high-school biology programs.
Sponsored Content:
SynDaver Products
SynDaver Labs products are made from a variety of materials including water, fibers and salts. Each of the tissues produced from these materials are required to be evaluated and validated for mechanical, chemical, thermal and dielectric properties (against the relevant living tissue). The company’s extensive validation helps to ensure that all simulated muscles, tendons, veins, arteries, nerves and organs (made from complex composites) are able to accurately mimic the properties of the discrete living tissues.
These life-like products developed by SynDaver Labs have been known to be used in a variety of healthcare fields. For example, surgical training, medical device testing, consumer products evaluation and ballistics testing programs have all employed the use of SynDaver Labs products.
Through the sale of these medical supplies and cast trainers, and by providing synthetic cadavers, Syndraver is able to focus business efforts on the development of synthetic human tissues for use in medical device verification and validation tests. While the company has made significant progress in their product development, the ultimate goal of SynDaver Labs is to be able to replace live animals and human cadavers. These replacements will have vast impacts on medical education and training with synthetic analogs which are more cost-effective than the relevant animal or human model.
Click Here to Connect to Leading Healthcare Simulation Vendors Like SynDaver
Through the push for these developments, SynDaver is simultaneously in the process of increasing the number of tissues in the company’s library, expanding the body of live tissue data upon which these materials are based and reducing the overall cost of the technology. The company is also working to develop a family of synthetic humans which breathe, bleed and react to stimulus with autonomy – some purely synthetic and some with living cells.
SynDaver Technology
The work which has become SynDaver Labs technology first began at the University of Florida over two decades ago, in 1993. The company’s initial studies involved the manufacture of synthetic trachea models. The purpose of these models were to replace live animals in the testing of airway devices and the development of interpenetrating polymer-fiber networks to mimic lumenal structures (such as vessels). Again, this early simulation has helped make healthcare education safer and more effective.
That being said, today, there are now materials which SynDaver Labs develops that are being used extensively in the industry as simple vein and artery mimics. SynDaver is constantly refining technologies, seeking new markets for existing products and designing new products for emerging applications. The implication of these products continues to become increasingly widespread as their purpose and success is demonstrated.
Back in 2017, SynDaver Labs further launched the SynDaver Canine to extend this application to the veterinary medical education community. Then, in 2018, SynDaver Labs also launched “CopyCat” (a synthetic feline designed for middle and high-school biology programs).
Also in 2018, and following beta-testing, SynDaver Labs announced a mass-market rollout of the SynDaver Advanced Modular Mannequin (SAMM), an open-sourced modular mannequin that comes with design plans and a 3D printer to allow the end-user to modify the mannequin according to the consumer’s own desires. You can see SynDaver’s products at the SSH IMSH meeting.
Further producing advanced technologies, SynDaver is nearing launch of the third-generation (3G) version of the SynDaver Synthetic Human and is also developing newborn, preemie and pediatric versions of the synthetic human product line. These models will enable surgical training in the neonatal and pediatric space in a way that is currently not possible without ever risking an actual human life.
The company is also developing military-specific versions of our surgical canine model which work well for surgical simulation programs. The standard version is permanently in use at the University of Florida, Texas A&M, the University of Saskatchewan and many other schools with simulation centers.
In these educational settings where moulage is regularly used to improve simulator presentation, the versions have completely replaced the use of both live dogs and canine cadavers in surgical education programs. SynDaver Labs is now working on additional animal models including cat, horse and rabbit. Also in development are human models that incorporate artificial intelligence to enable interaction with live humans.
SynDaver Labs Latest News

Global Healthcare Simulation News Update January 2024

11 More Key Terms to Help Your Healthcare Simulation Program Develop Faster

IMSH 2018 Starts with a Bang — SynDaver Acquires Lifecast Body Simulation

SynDaver Labs Gives Away $1.5 million in Stock to Employee for Christmas
Sponsored Content: